

Two recent chemical incidents at the controversial Silfab Solar factory in Fort Mill have brought renewed attention to the complex manufacturing processes planned at the site.
Earlier this month, York County reported that 1,530 gallons of potassium hydroxide spilled into the retention pond by Flint Hill Elementary School, triggering a major emergency hazmat. Silfab later pumped out 300,000 gallons of liquid from the pond to be disposed of off site, according to DES.
Days later, officials confirmed a second leak involving hydrofluoric acid, prompting Flint Hill Elementary to close for 2 days and the South Carolina Department of Environmental Services to order the company to pause operations while an investigation takes place.

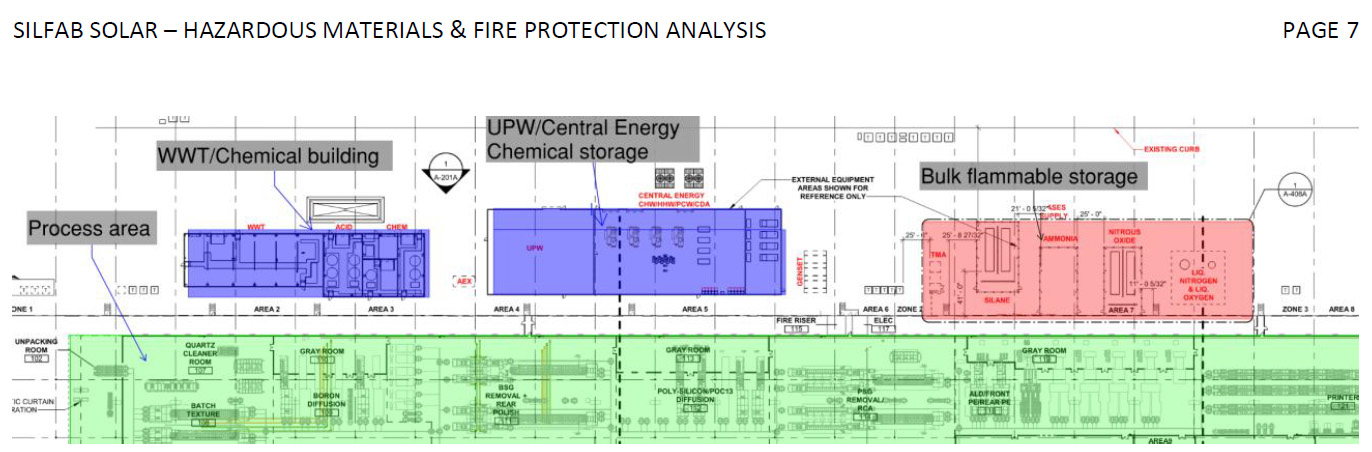
Those incidents have raised broader questions about what exactly happens inside Silfab’s experimental 900,000 square foot TOPCON solar cell factory (which would become one of the largest TOPCON cell factories in the world if completed).
According to three technical documents submitted for the Fort Mill plant – including the facility’s Hazardous Materials Inventory System (HMIS) report, DES Air Permit, and Industrial Wastewater Treatment Plans — the factory is designed to transform raw silicon wafers into finished solar panels through a multi-stage industrial process involving strong acids, explosive gases, and chemical wastewater treatment systems.
An Overview of the Manufacturing Process
Solar cell production follows a series of transformations that gradually turn slices of silicon wafers into complex mini-power plants.
The stages include:
-
Grey cell manufacturing
-
Blue cell manufacturing
-
Finished solar cell manufacturing
-
Finished panel (module) manufacturing
Each step alters the wafer’s structure using chemical reactions, high-temperature furnaces, and thin-film deposition systems.
By the end of the process, dozens of completed cells are connected together using high heat and chemicals into a finished solar panel (module) capable of generating electricity from sunlight.
However, the manufacturing chain also produces air emissions, chemical wastewater, and industrial sludge, which Silfab plans on reducing through pollution control equipment, onsite treatment systems, and offsite sludge dumping.
1. Wafer Manufacturing → Grey Cell
The process begins with silicon wafers, thin slices of purified crystalline silicon typically 200–300 micrometers thick.
Manufacturing steps
-
Chemical cleaning
-
Surface texturing
-
Semiconductor doping
-
Etching
Chemicals used
-
Hydrofluoric acid (HF)
-
Hydrochloric acid (HCl)
-
Potassium hydroxide (KOH)
-
Nitric acid (HNO₃)
-
Boron Trichloride (BCL3)
High-temperature diffusion furnaces around 1000°C create the electrical p-n junction that allows the cell to generate electricity.
Waste produced
-
Acidic wastewater
-
Fluoride-containing wastewater
-
Silicon sludge
These wastes are typically treated to form calcium fluoride sludge for disposal.
At the end of this stage, the wafer becomes a grey cell.
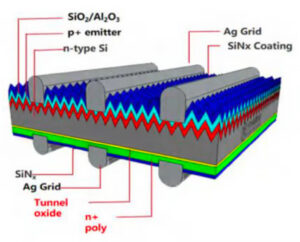
2. Grey Cell Manufacturing → Blue Cell
 The grey cell then receives several thin coatings that improve electrical performance.
The grey cell then receives several thin coatings that improve electrical performance.
Manufacturing steps
-
Tunnel oxide layer formation
-
Polycrystalline silicon deposition
-
Surface passivation
-
Anti-reflective coating
The tunnel oxide layer is only 1–2 nanometers thick, while the poly-silicon layer is about 60–100 nanometers thick.
Chemicals and gases used
-
Silane (SiH₄)
-
Ammonia (NH₃)
-
Nitrogen carrier gases
-
Aluminum oxide compounds
These coatings are typically applied using PECVD or LPCVD deposition systems.
Waste produced
-
Process gas exhaust
-
Nitrogen oxide emissions
-
Fine silicon particles
After the anti-reflective coating is applied, the device becomes a blue cell.
3. Blue Cell Manufacturing → Finished Cell
 The next stage adds electrical contacts that allow current to flow out of the cell.
The next stage adds electrical contacts that allow current to flow out of the cell.
Manufacturing steps
-
Screen printing conductive pastes
-
Infrared drying
-
High-temperature firing
Materials used
-
Silver paste
-
Aluminum paste
-
Organic solvents
Waste produced
-
Volatile organic compounds (VOCs)
-
Metal particulates
-
Furnace exhaust gases
After metallization is complete, the device becomes a finished solar cell.
4. Finished Cell Manufacturing → Finished Panel
The final stage assembles finished solar cells into a complete solar module.
Manufacturing steps
-
Cells are soldered together with copper ribbons to form strings
-
Encapsulation layers are placed above and below the cells
-
The assembly is laminated under heat and vacuum
-
A polymer backsheet, aluminum frame, and electrical junction box are installed
Chemicals and materials used
In addition to structural materials, this stage uses several chemical components:
-
EVA (ethylene-vinyl acetate) encapsulant sheets
-
Polymer backsheets (often PET-based laminates)
-
Silicone or polyurethane sealants
-
Flux chemicals and potting compounds
VOC emissions produced
During the high-heat lamination and curing process, several materials can release volatile organic compounds (VOCs).
Wastewater and Fluoride Sludge

Silfab’s factory would use over 1.2 million gallons of water per day and would generate significant volumes of chemical wastewater.
According to Silfab’s wastewater treatment design documents, the plant is expected to treat wastewater containing:
-
hydrofluoric acid
-
potassium hydroxide
-
hydrochloric acid
-
metal residues
-
dissolved silicon
Treatment process
The wastewater system neutralizes acids and bases and removes fluoride through precipitation reactions.
Hydrofluoric acid reacts with calcium compounds to form calcium fluoride sludge, which must be collected and disposed of as industrial waste.
Can Silfab Legally Operate in “LI”?
There is a growing number of Fort Mill residents, now numbering in the thousands, calling for the facility to move to a different location given its size, chemical usage, and the pollution and waste it would create.
Move Silfab organizers argue the facility is operating in a zoning district that does not allow the type of hazardous materials planned for the site. York County’s zoning code states that “Light Industrial does NOT include hazardous material treatment or storage facilities.” (York County Code §155.1301).
They also point to other provisions in the county’s zoning law. Under the code, “the Zoning Administrator must issue formal zoning compliance approval before permits are valid.” (York County Code §§155.1122–1123). In spite of numerous FOIA requests and private meetings, York County officials have still never produced a signed zoning compliance letter for Silfab’s operations – without one, all of Silfab’s permits and their Certificate of Occupancy would be legally void (according to SC Peterson v. Greenville County, 2011, “An invalid zoning permit issued in violation of applicable zoning laws confers no rights and is void ab initio”).
The Attorney General of South Carolina, Alan Wilson, has also confirmed that the York County Council has the authority to revoke Silfab’s CO if they do not have proper zoning approvals.
Move Silfab is now organizing a mass protest on Monday night, calling for the York County Council to take a formal vote on the project.


